DESCRIPTION
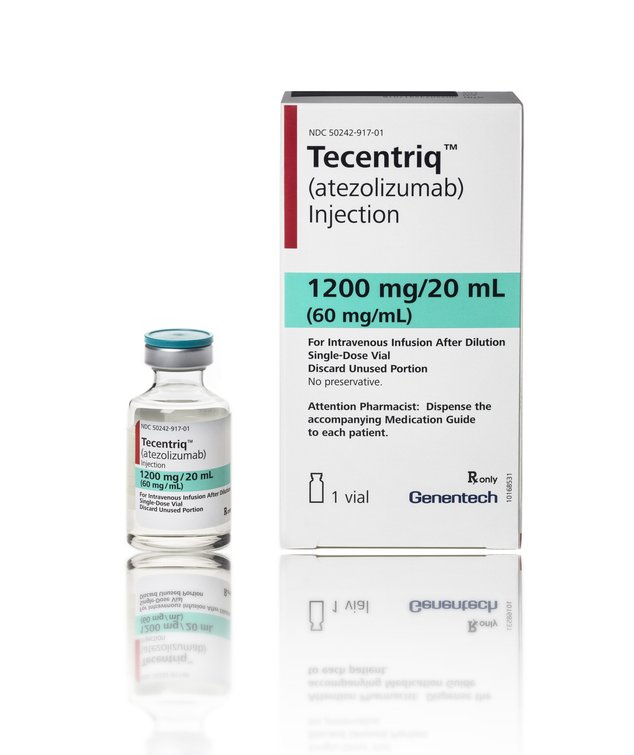
TECENTRIQ 1200mg (Atezolizumab) is a cancer immunotherapy designed to work with the body’s own immune system. TECENTRIQ is the first and only approved anti-PDL1 medicine for patients with specific types of advanced urothelial carcinoma and non-small cell lung cancer.
Manufacturer : Genentech, Inc.
ADDITIONAL INFORMATION
Strengths available : 1200 mg/ 20 ml
Storage : Store at Temperature between 2°C-8°C. Do not freeze.
Drug Type : Anti-PD-L1 monoclonal antibody
Dosage : Recommended Dosing –
The recommended dose of TECENTRIQ is 1200 mg administered as an intravenous infusion over 60 minutes every 3 weeks until disease progression or unacceptable toxicity. If the first infusion is tolerated, all subsequent infusions may be delivered over 30 minutes. Do not administer TECENTRIQ as an intravenous push or bolus.
SIDE EFFECTS
Most common side effects are Decreased appetite, Nausea, Urinary tract or other infection, Constipation, Fever, Diarrhea, Colitis (bowel inflammation), Swelling, Abdominal pain, Vomiting, Shortness of breath and/or cough and/or wheezing, Neck pain/back pain, Rash and/or itching, Hematuria (blood in the urine), Arthralgia (joint pain), Hyponatremia (low sodium level), Thyroid changes, Autoimmune reactions* and infusion reactions.
PACK SIZE
Pack of 1 Vial
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of TECENTRIQ Atezolizumab 1200 mg Injection based in India - To buy TECENTRIQ Atezolizumab 1200 mg Injection or know its cost price contact us here.
We supply & sell TECENTRIQ Atezolizumab 1200 mg Injection for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.