DESCRIPTION
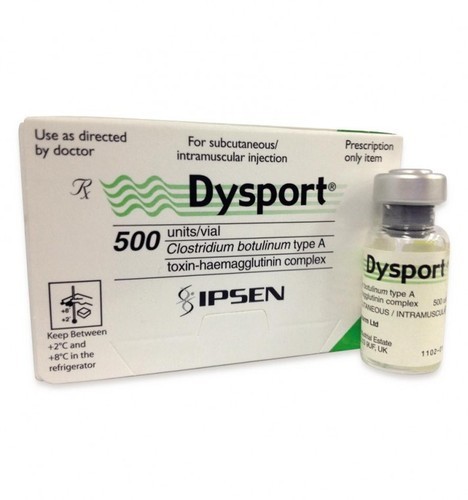
Dysport is used to treat cervical dystonia, it blocks nerve activity in the muscles, causing a temporary reduction in the muscle activity. It is also used to temporarily lessen the appearance of facial wrinkles such as those that form between eyebrows, on the forehead and around the eyes.
Manufacturer : Biopharmaceuticals Inc Ipsen
ADDITIONAL INFORMATION
Strengths available : 500 units
Form : Injection
Storage : Store at 2 °C to 8°C
Is the medicine FDA approved : Yes
Date of Approval : April 29, 2009
Dosage : The recommended initial dose of DYSPORT® for the treatment of cervical dystonia is 500 Units given intramuscularly as a divided dose among affected muscles in patients with or without a history of prior treatment with botulinum toxin. Limiting the dose injected into the sternocleidomastoid muscle may reduce the occurrence of dysphagia.
SIDE EFFECTS
More Common side effects are difficulty with swallowing or speaking, hoarseness, muscle or bone pain, muscle weakness, seizures, sore throat, voice changes, Less Common, Blistering, burning, crusting, dryness, or flaking of the skin, body aches or pain, chills, cough, cough producing mucus, diarrhea, difficult or labored breathing, ear congestion, fever, general feeling of discomfort or illness, headache, itching, scaling, severe redness, soreness, or swelling of the skin, joint pain, loss of appetite, loss of voice, muscle aches and pains, nausea, shivering, sneezing, stiff muscles, stuffy or runny nose, sweating, tightness in the chest, trouble sleeping, unusual tiredness or weakness, vomiting.
PACK SIZE
Pack of 1 vial
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of Dysport Abobotulinumtoxin A 500 units based in India - To buy Dysport Abobotulinumtoxin A 500 units or know its cost price contact us here.
We supply & sell Dysport Abobotulinumtoxin A 500 units for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.