Description
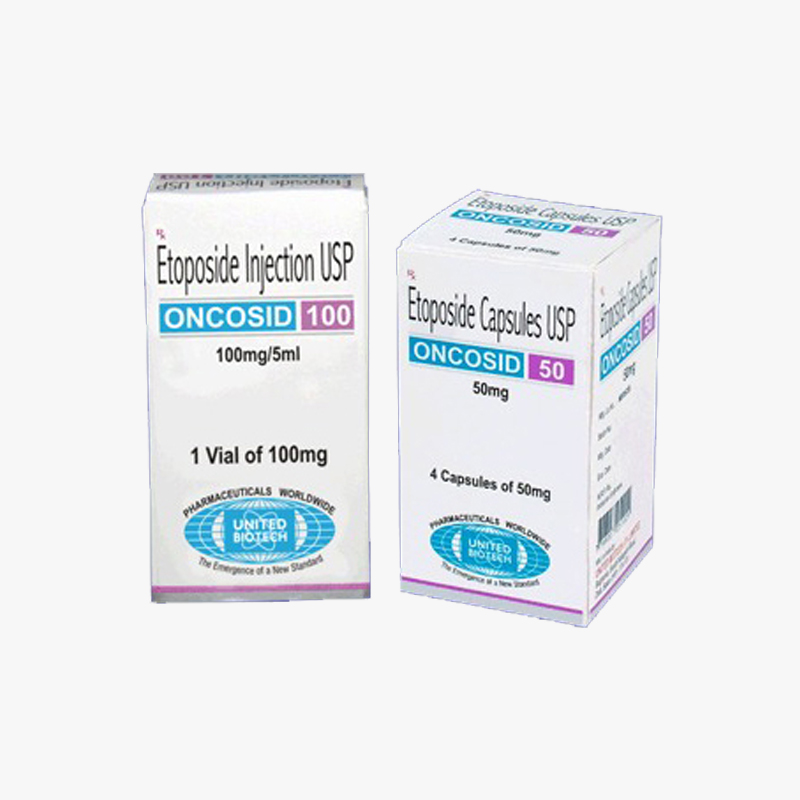
Etoposide Injection USP 100 mg Etoposide (also commonly known as VP-16) is a semisynthetic derivative of podophyllotoxin used in the treatment of certain neoplastic diseases. It is 4′-demethylepipodophyllotoxin 9-[4,6-O-(R)-ethylidene-β-D-glucopyranoside].Etoposide Injection USP is available for intravenous use as 20 mg/mL solution in 100 mg (5 mL),Etoposide Injection USP is indicated in the management of the following neoplasms:
- Refractory Testicular Tumors
- Small Cell Lung Cance3s corporation is Supplier,Exporter ,Wholesaler for Etoposide Injection USP 100 mg in India.
We even offer contract manufacturing services from WHO GMP approved factories.
Additional Information
Side Effects
Pack Size
3S Corporation is a WHO GDSP approved & licensed Pharmaceutical stockist/wholesaler/distributor/exporter/importer of Etoposide Injection USP 100 mg based in India - To buy Etoposide Injection USP 100 mg or know its cost price contact us here.
We supply & sell Etoposide Injection USP 100 mg for Reference Listed Drugs, Government Tenders, Shortage Lists, Emergency Imports, Name Patient Drugs, Comparator Drug Studies, Un-licensed Importation, clinical trial samples & Bio-Similars.